Telix FDA Approval Strengthens Radiopharma in Indy
Subscriber Benefit
As a subscriber you can listen to articles at work, in the car, or while you work out. Subscribe Now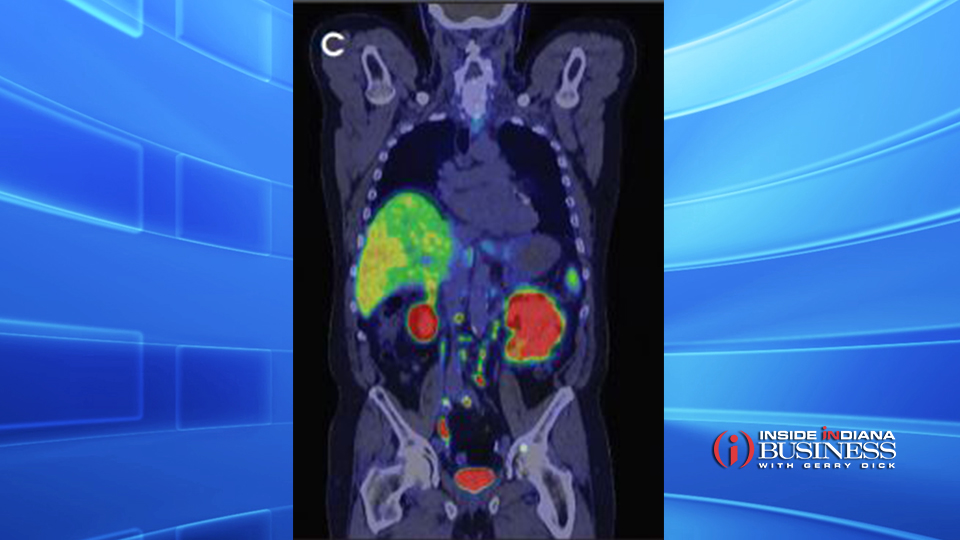
Industry experts say Indiana is emerging as a powerful hub for nuclear medicine, and the most recent fuel to be added to the fire is Telix Pharmaceuticals earning approval from the U.S. Food and Drug Administration to launch its first commercial product. The radiopharmaceuticals sector, also known as nuclear medicine, is gathering momentum in central Indiana; companies have committed to investing $200 million in new facilities by the end of 2023. Calling Fishers home since 2020, Telix says it will leverage Indy’s distribution might to break through barriers that have limited how many patients can access cutting-edge imaging tools for prostate cancer.
Nuclear medicine is a specialized area of radiology that uses very small amounts of radioactive materials, or radiopharmaceuticals, to diagnose or treat disease. Showing great promise, the commercial critical mass is now building; a recent industry report predicts the global market for nuclear medicine will increase from $5 billion to $14 billion by 2025.
With locations in Australia, Belgium and Japan, Telix opened its Fishers facility in 2020 to operate as its U.S. headquarters—and growing even faster than anticipated—expects to move to an even larger location in the city soon. Telix Head of Marketing for the Americas Jason Kaufmann says the recent FDA approval is another significant boost to business, signaling that the company “is a serious player in nuclear medicine.”
Telix specializes in Molecularly Targeted Radiation, and its first product, called Illuccix, relies on this “lock and key” concept.
“You have a targeting agent, which can be a small molecule or antibody that is attracted to particular types of cell; in the case of [Illuccix], it’s to prostate cancer cells,” says Kaufmann. “That particular targeting agent is attracted to that cancer cell; then we attach a nuclear element to that targeting agent. So when [the targeting agent] is injected into the patient, it circulates and is drawn to those cancer cells and only to those cancer cells, in general.”
The name Illuccix alludes to its “illuminating” ability; the technology is used in PET scans to determine if the cancerous cells have spread to areas outside the prostate, and if so, identify their precise location. A “payload” can be attached to the targeting agent; a gallium-based radioactive isotope is the payload for Illuccix, causing cancer cells to “light up,” and be visible in PET scans.
Kaufmann says these PET scans—powered by nuclear medicine—to identify where prostate cancer has metastasized (spread) are more effective than the conventional methods of bone and CT scans. However, nuclear medicine can be more challenging to deliver to patients; first, the production of radiopharmaceuticals demands mind-boggling manufacturing facilities. A cyclotron, for example, produces radioactive isotopes; the single piece of machinery costs in the millions and is comprised of enormous magnets that weigh more than 100 tons.
A second major challenge is that, due to the radioactive materials in nuclear medicines, the drugs have an extremely short shelf life, which can limit how far the product can travel from the place where it’s manufactured. Illuccix, for example, has only a four-hour shelf life after it’s produced, and that’s why Telix is partnering with PharmaLogic and Cardinal Health to help clear the distribution hurdle.
Ohio-based Cardinal Health has a major presence in central Indiana. Telix produces the Illuccix “kit” with the base components, then Cardinal Health adds the nuclear element and uses its large distribution network to beat the four-hour clock.
“It’s a challenge,” says Kaufmann, “and that’s one reason we’ve partnered with Cardinal Health. With the sheer number of their locations and number of gallium generators they have across the U.S.—as well as PharmaLogic’s, another distribution partner—we have such wide coverage. That mitigates the shelf life challenge.”
Through the partnerships, Telix says it will be able to provide Illuccix to more than 85% of eligible PET imaging sites throughout the U.S.
Kaufmann believes the company is poised for even more growth in central Indiana. It currently uses an out-of-state contract manufacturer, but anticipates adding production in the Hoosier state, and Telix will likely double its current U.S. workforce of 70 by 2023.
“As a startup and healthcare company, we’ve always said that we wanted to help patients; that’s been our mantra and one of our core beliefs since we started,” says Kaufmann. “And now [with FDA approval], we can actually fulfill that dream and really start making a difference in patients’ lives on a scale we’ve never been able to before.”
For full Prescribing Information and Important Safety Information, visit www.illuccix.com
Kaufmann says Telix’s pipeline includes an imaging agent for renal (kidney) cancer and glioblastoma (brain cancer).
Kaufmann says, because radiopharmaceuticals have such a short shelf life, it’s clear why nuclear medicine is exploding in central Indiana.
