South Bend Hospitals Test Notre Dame-Developed Breast Imaging Device
Subscriber Benefit
As a subscriber you can listen to articles at work, in the car, or while you work out. Subscribe Now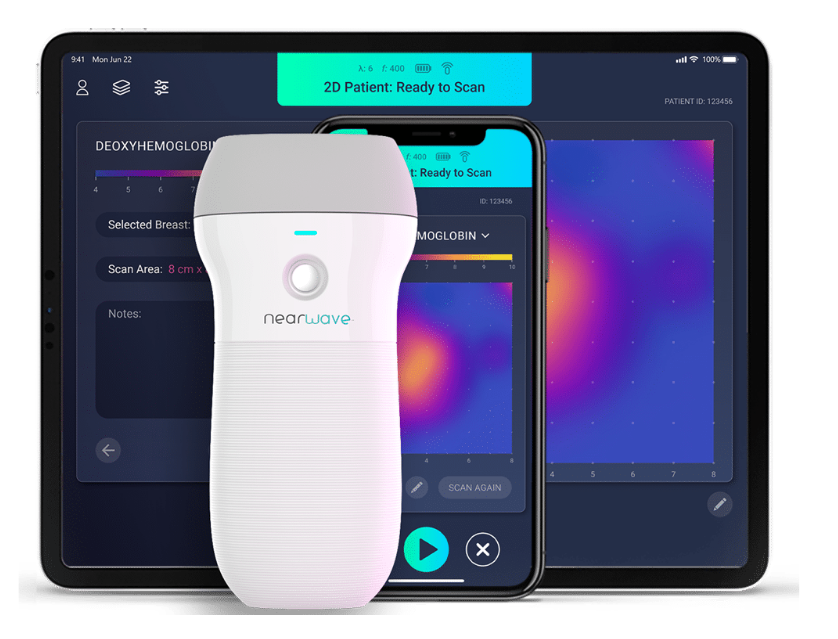
What started as a PhD project for a University of Notre Dame student has exploded into a “billion-dollar idea” that could transform imaging standards for breast cancer treatment. Called NearWave Imager, the handheld device will help doctors determine if a certain treatment is meeting its goal to shrink or kill a tumor. It’s an answer that doctors currently must wait months for, but the NearWave device can deliver it within one week, says Chief Executive Officer Dr. Roy Stillwell. About 60 Hoosier women in the South Bend area are now part of a study to test the device—getting newfound clarity about how best to fight their cancer.
The first—and perhaps most disruptive—application for the NearWave Imager is for neoadjuvant chemotherapy, which is treatment given to shrink, kill, or stop the growth of a tumor before the patient undergoes surgery, such as a lumpectomy or mastectomy.
“About 30% of women don’t respond to the primary therapy during that pre-surgery therapy period,” says Stillwell, who is also chief scientist. “For three to six months, they’re taking really nasty chemotherapy that’s making them sick, and there may be no effect [on the tumor].”
Currently, there are no imaging systems to monitor a patient’s response to neoadjuvant chemotherapy, so doctors are unable to see for several months if their efforts are successfully shrinking or killing the tumor. However, NearWave says its handheld imaging device can measure the amount of blood, water and lipid (fat) in the area of the tumor—ultimately, determining if it’s responding to the chemotherapy.
“If you’re not responding, then [doctors] can move you to a different therapy really fast, rather than waiting six months to find out if you’re responding,” says Stillwell. “We can tell you within a week, which is huge in the oncology space—that’s what everyone has wanted forever—to tell whether or not therapies are working.”
The device, which won the Mira Award for Innovation of the Year in 2021, centers on the power of near-infrared spectroscopy (laser light). NearWave co-founder and Notre Dame Electrical Engineering Assistant Professor Dr. Thomas O’Sullivan says physicists and engineers have long understood the potential of “laser” technology but simplifying it for clinical use was a hurdle too high. Based on discoveries made in O’Sullivan’s lab, NearWave says it’s cracked the code.
The handheld NearWave Imager beams several light sources—lasers, in basic terms—at the area of concern, and based on how blood, water and fat absorb the light, doctors can get a much clearer picture.
“If there’s a tumor, there’s often a displacement of fat; fat gets out of the way because a tumor is growing, so there’s a drop in fat (lipid),” says Stillwell. “We can see how much blood is in a certain area; malignant tumors, for example, have much, much more blood in the surrounding area [versus benign].”
A study is now underway in South Bend at the Paqui and Brian Kelly Comprehensive Breast Center at Saint Joseph Health System, with a second hospital expected to join soon.
“It absolutely could be a disruptive technology,” says Stillwell. “There are lots of landmines bringing a medical device to market: you have to get through the FDA in a really specific way; to get it to practical use, you have to have a reimbursement strategy for the doctors. There are a lot of pieces that have to be in place—we have a bunch of experts helping us with that. It’s potentially a billion-dollar idea if we can make sure we avoid all the landmines on the way there.”
The startup worked with the Notre Dame IDEA Center in the company’s earliest stages and is now working to close its initial seed round; NearWave says it should have the $1.9 million round “nailed down” this summer. The startup will submit the device for FDA approval this year, launch additional studies soon and fine-tune the device’s design, so it can be mass manufactured.
“When [the device]…is actually being used in the clinic, that first patient who gets a life-saving treatment because we were able to switch them to the right therapy within a couple weeks—that’s what I’m most excited about,” says Stillwell. “It only has to be one patient. [NearWave] is likely [to help] many thousands of people—but all of this work we’re doing, all of the sleepless nights—to save the life of just one person is totally worth it.”
Stillwell says there are two other very promising breast cancer applications for the device: switching treatments quickly for patients with triple negative breast cancer, and improved mammography for women with dense breast tissue.
Stillwell says the study underway at the South Bend hospitals involves using the handheld device to determine if a tumor in the breast is malignant or benign.
