Fishers biotech firm lands additional FDA approval
Subscriber Benefit
As a subscriber you can listen to articles at work, in the car, or while you work out. Subscribe Now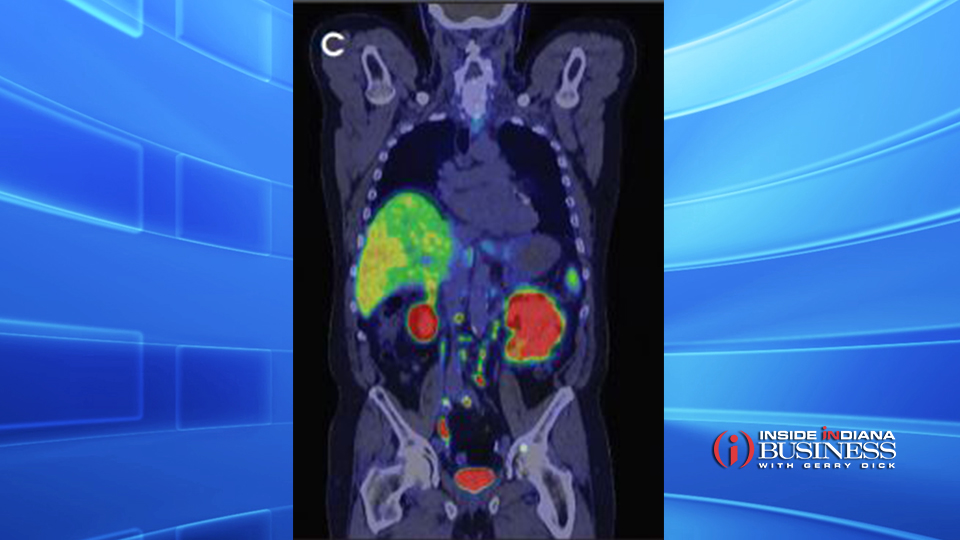
An Australian biotech company that has its U.S. headquarters in Fishers has received additional approval from the U.S. Food and Drug Administration. Telix Pharmaceuticals says its lead prostate cancer imaging product, Illuccix, is now authorized to select patients who may be eligible for another FDA-approved therapy developed in Indiana.
Illuccix was first approved by the FDA in December 2021. The imaging agent is designed to target and “light up” cancerous cells, making it easier for oncologists to determine through a PET scan if the cells have spread to areas outside the prostate and if so, identify their precise location.
The FDA has approved a supplementary New Drug Application for Illuccix to select patients with metastatic prostate cancer who are candidates for Pluvicto, the only FDA-approved radioligand therapy for prostate cancer.
“This additional indication further demonstrates our continued commitment to support patients fighting prostate cancer and to empower the doctors who treat them,” Telix Americas CEO Kevin Richardson said in written remarks. “Clinicians now have the ability to use Illuccix in more stages of the patient journey, to confidently and accurately detect and help manage this disease.”
Telix generated $50.5 million in revenue from sales of Illuccix in the fourth quarter of 2022, a 43% increase from the third quarter.
Pluvicto received its FDA approval in March 2022. Telix said an estimated 32,000 U.S. patients may be considered for the therapy.
The drug was developed by Dr. Philip Low, the Presidential Scholar for Drug Discovery at Purdue University. Pluvicto is being sold by Novartis, which acquired Low’s Purdue-based startup, Endocyte, in 2018 in a $2 billion deal.
Telix Pharmaceuticals moved into its new Fishers headquarters inside the Crew CarWash HQ building near Interstate 69 and East 116th Street last October. The company employs about 100 U.S. employees, 33 of which are located in Fishers.
You can read more about Telix and the rise of nuclear medicine in this April 2022 article by the IBJ’s John Russell.
